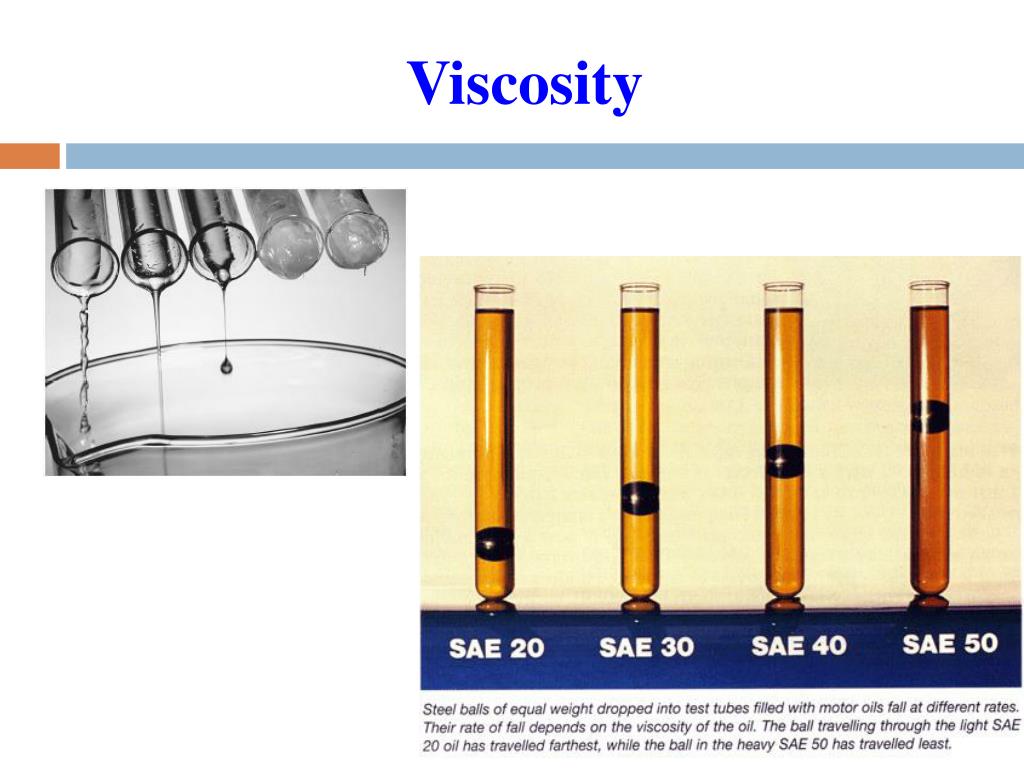
The video below shows several different long chained oils, each progressively more viscous. Glycerol, CH 2OHCHOHCH 2OH, is viscous partly because of the length of the chain but also because of the extensive possibilities for hydrogen bonding between the molecules. Fuel oil, lubricating grease, and other long-chain alkane molecules are quite viscous for this reason. 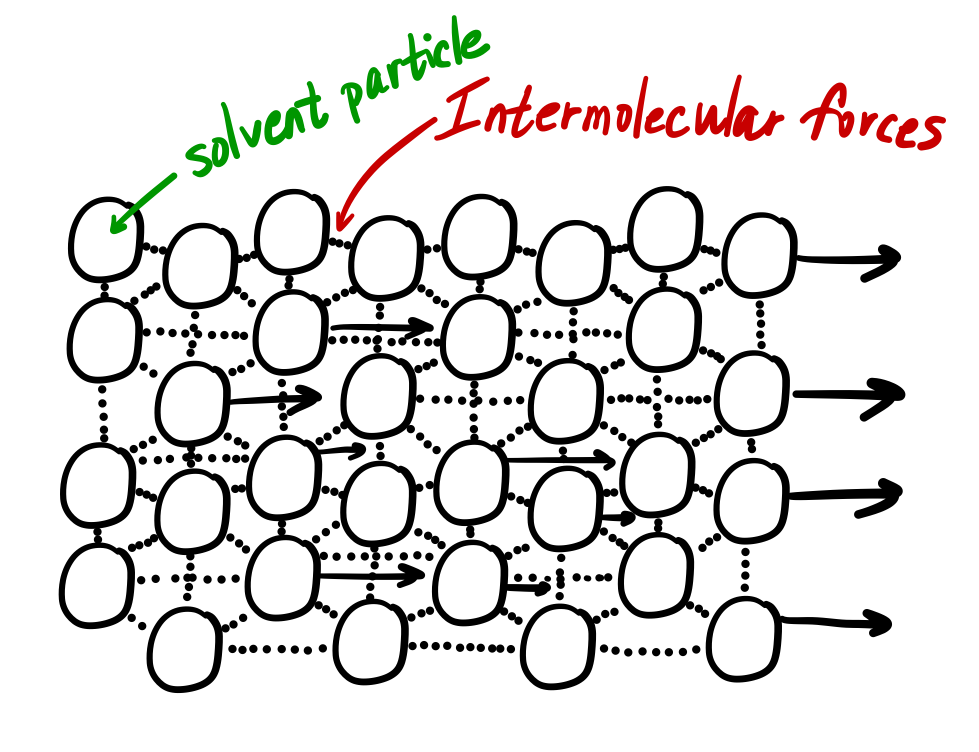
This is because the molecular chains get tangled up in each other like spaghetti-in order for the liquid to flow, the molecules must first unravel. Liquids containing long molecules are invariably very viscous. Honey, mostly glucose and fructose (see image below) is a good example of a liquid which owes its viscosity to hydrogen bonding. Liquids whose molecules are polar or can form hydrogen bonds are usually more viscous than similar nonpolar substances. Viscosity is governed by the strength of intermolecular forces and especially by the shapes of the molecules of a liquid. Those like ether or gasoline which flow very readily have low viscosities. According to the above mathematical formula, viscosity is defined as the force per unit area originating between the two layers moving with the unit velocity. Liquids which flow very slowly, like glycerin or honey, have high viscosities. The resistance to such flow is called the viscosity. The viscosity of a liquid decreases with increase in temperature.\)īecause its molecules can slide around each other, a liquid has the ability to flow. With increase in temperature, the kinetic energy of the molecules of the liquid increases which can overcome the intermolecular forces. Hydrogen bonding and van der waals forces are strong enough to result into high viscosity of the liquid. Water has higher viscosity than methyl alcohol because intermolecular forces in water are greater than those in methyl alcohol. Greater are the intermolecular forces, higher is the viscosity of the liquid. The units of viscosity are dynes sec cm -2.This quantity is called 1 Poise.ġ poise = 1 g cm -1 s-1 = 0.1 kg m -1 s -1Ģ) Effect of nature of the liquid on viscosity  
This force of friction which one part of the liquid offers to another part of the liquid is called viscosity.Ĭoefficient of viscosity may be defined as the force of friction required to maintain a velocity difference of 1 cm/sec between two parallel layers, 1 cm apart and each having an area of 1 sq cm. In other words, every layer offers some resistance or friction to the layer immediately below it. This type of flow in which there is a regular gradation of velocity in going from one layer to next is called laminarĪs we move from centre towards the walls, the velocity of the layers keep on decreasing. As we move from the wall towards the centre of the tube, the velocity of the cylindrical layers keeps on increasing till it is maximum at the centre. The layers which is in contact with the walls of the tube is almost stationary. Imagine the liquid to be made up of a large number of thin cylindrical coaxial layers. All parts of the liquids do not move through the tube with same velocity. The liquids which flow rapidly have low internal resistance which is due to weak intermolecular forces and hence are said to be less viscous or said to have low viscosity.Ĭonsider a liquid flowing through a narrow tube. The liquids which flow slowly, have high internal resistance which is due to strong intermolecular forces and therefore, are said to be more viscous or are said to have high viscosity. This internal resistance to flow possessed by a liquid is called its viscosity Some liquids like water, ether flow rapidly why some other liquids like glycerine, castor oil flow quite slowly.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
